A TIMELINE FOR
THE PLANET click for Home Page
The dawn of Life
Astronomers believe that Life’s building blocks winged
in from Outer Space. But they would
claim that wouldn’t they.
Enzymes When did life emerge? Phospholipids How did Life emerge?
The astronomers have detected many of the building
blocks of life in the tenuous atmospheres of stars. But earth-bound scientists have found plenty
of places here on Earth that could have given emerging life its first leg up
just as easily. Many of Life’s building
blocks do seem to be incredibly easy to make.
It’s noticeable, as we’ve hinted, that the astronomers
are the main exponents of the theory that our planet’s life originated in outer
space. And some would certainly argue
that their motivation is plain old-fashioned empire building.
Whichever theory takes your fancy, the difficult bit
remained to be done here on Earth. For
other building blocks are much harder to make.
As we’ll see later, scientists are
still struggling to understand how emerging life managed to make the jump from
the simple building blocks to something reasonably described as ‘life’.
All life the same
One of the things that originally got me following
this story was a number of questions like the following. “How is it that we humans can eat, and obtain
sustenance from, virtually every kind of living organism on the planet?” There are exceptions of course, mainly those
organisms that have invested valuable resources in making poisons. They don’t do this for fun. They do it specifically to avoid being eaten.
And “why is it that biologists comb the tropical
rainforests, looking for vaccines and other chemicals which might help cure
humans?”
The answer turned out to be very simple. Down at the basic chemical level, all life is
exactly the same.
Proteins and the
genetic code
We’ve now reached the point where the scientists begin
to be on firmer ground. The following is
fact.
With rare exceptions: all life uses the same 5
so-called ‘bases’ to make its DNA and RNA with.
(It’s 5 because RNA uses one that’s different from its DNA
equivalent.) Life also uses the same 20
‘amino acids’ to make its proteins from.
And, again with rare exceptions, it uses the same genetic code to
translate between the two.
 The
next couple of paragraphs are a bit turgid.
I’ve put them in to lend an air of “artistic verisimilitude to an
otherwise bald and unconvincinv narrative” (W.S. Gilbert, ‘The Mikado’). Please feel free to skip them.
The
next couple of paragraphs are a bit turgid.
I’ve put them in to lend an air of “artistic verisimilitude to an
otherwise bald and unconvincinv narrative” (W.S. Gilbert, ‘The Mikado’). Please feel free to skip them.
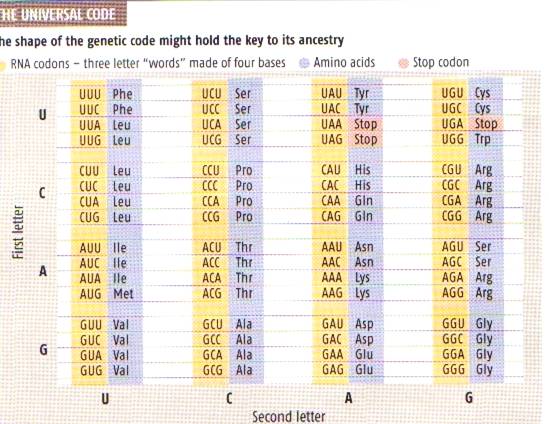
This diagram of Life’s genetic code comes from New Scientist (13.8.05). The yellow column indicates a ‘word’, or
codon in the jargon, made up of three of the 4 available ‘bases’. If you want to know more about bases, I’m
afraid you will have to look them up on the Internet. The blue column shows which of the 20 amino
acids the codon specifies. The second
diagram indicates these. It comes from Scientific American (Oct. 85).
The first thing to notice is the large amount of
redundancy in the code. A code of 3
letters, selected from 4 different possibilities, gives us 64 possible amino
acids. And yet life has only found use
for 20. In many of the codons, the third
letter is ignored. And evolutionary
biologists have found something queer about the way first letter is used too
(which I don’t understand and won’t attempt to explain).
This has led them to speculate that the first code
only used the middle letter of the three.
The outside letters were simply used as ‘spacers’. This implies that the first organisms that
used the code had to make do with 4 different amino acids. This was very limiting and, to get a wider
choice, the outside letters were gradually brought into play. Presumably emerging life only found use for
20 amino acids. Or maybe there are only
20 that can be specified in this way.
This diagram shows the 20 amino acids that life chose. I won’t attempt to explain it, but it’s laid
out to show how easily 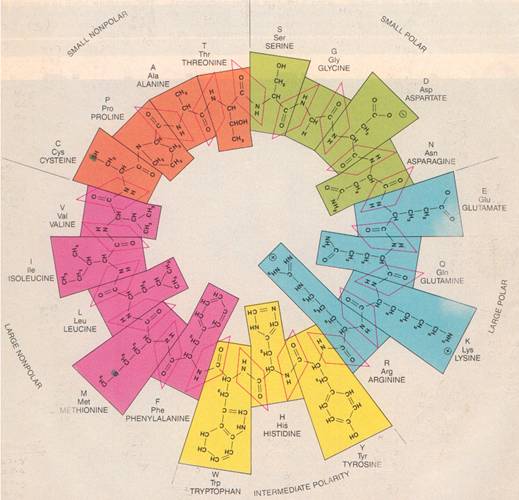 they
link together. What may be less obvious
is how similar the central portions are.
The differences lie mainly in the bits that branch off.
they
link together. What may be less obvious
is how similar the central portions are.
The differences lie mainly in the bits that branch off.
Please start paying attention again now.
Quite quickly emerging life ended up with the code we
find today. I say ‘quite quickly’ because
the code must have become fixed before any
of life’s surviving branches split off.
The amino acids do most of their work by linking
together into long chains – to make proteins.
Proteins are used to make structural components. For these the chains are straight. And many different chains have to link
together, side by side, to provide stiffness.
It’s a bit like dipping a piece of string into molten wax. It goes in flexible and comes out stiff.
Enzymes
The other main things that proteins make are
enzymes. Enzymes do a different
job. They are the cell’s chemical
factories. Enzymes take in small
chemical building blocks, and snap them together to make larger ones. For this, they have to clump together into a
ball. And it’s the precise shape into
which they clump that gives each individual enzyme its unique properties.
The clump has to include little niches, that are just
the right size and shape to enable a particular molecular ‘building block’ to
slip into it. It’s vital that only the
right building block can fit in. Next
door there’s a niche for another building block; also of course exactly the
right one. The clump is continually
vibrating. It forces the two building
blocks together and pops them out before the next vibration can separate them
again.
Other enzymes break down the large building blocks, so
that the smaller blocks can be reused.
In this case of course the single niche is the right size for the
selected component to slip into. It is
snapped apart and the individual blocks are ejected before they can be
recombined.
Why have different enzymes for the two jobs? Presumably it’s because it provides better
control of the two opposite activities.
It’s the bases (adenine, cytosine etc.) that are
difficult to make. The amino acids are
much easier, and it’s these that astronomers tend to find on their patch.
When it comes to plants and animals, even fungi, the
similarities are very much greater still.
We are all based on the same eukaryotic’
cells. Indeed you would be hard put
to it to distinguish a plant cell from an animal or fungal cell, unless it was
a green one. Then it would contain chlorophyll for converting light into
energy.
Plant and animal genes, and probably those of fungi
too, tend to be remarkably similar. The
tropical rain forests are fundamentally some of the easiest places on the
planet to live, with constant warmth and moisture. This makes them the most competitive and
therefore paradoxically some of the toughest.
Everything that lives there has to work extra hard to avoid being eaten
and being infected by diseases. Since we
are so similar, there’s every chance that the protective chemicals that they
are forced to invent will be useful to us too.
When did Life emerge?
We will never know when Life first appeared on our
planet. If you take the view that
“extraordinary claims require extraordinary evidence” then you will want to put
the date fairly late. Even 3½ thousand
million years ago may be too early for you.
But if you follow the “absence of evidence is not evidence of absence”
way of thinking then you will want to put the dawn of life much earlier. You will, for example, be much more inclined
to believe that the controversial evidence for life in the Isua deposits is real.
The legendary Stanley Miller was once asked how long
it might have taken for life to originate.
He said “a decade is probably too short, and so is a century. But ten- or a hundred-thousand years is
probably O.K, and if you can’t do it in a million years then you probably can’t
do it at all”.
The advent of life was originally thought to be a
millions-to-one chance, and that we are probably unique in the Universe. But Miller is expressing very clearly the
opposite view – that if the conditions are right, then the emergence of life is
natural and probably inevitable. I
think this is a widely held view these days.
There’s evidence from zircons
that conditions could have been conducive to life as early as 4.4 thousand
million years. This is less than 200
million years of our planet’s formation.
There’s no evidence at all that life actually got going as incredibly as
early as this. Nor will there  ever
be. But it’s perfectly possible. Unfortunately there were some terrible
upheavals that happened after that. Some
scientists believe that these would have wiped out any life that did
emerge. However other scientists reckon
that the evidence points to these late upheavals being a good deal less
devastating than they have been painted.
For my part, I’ve always felt that it would have been incredibly hard
for an upheaval to completely wipe out every last vestige of life on the
planet. And a single small colony,
buried deep in the mud at the bottom of a deep ocean, would be enough to
re-colonise the planet as soon as conditions eased.
ever
be. But it’s perfectly possible. Unfortunately there were some terrible
upheavals that happened after that. Some
scientists believe that these would have wiped out any life that did
emerge. However other scientists reckon
that the evidence points to these late upheavals being a good deal less
devastating than they have been painted.
For my part, I’ve always felt that it would have been incredibly hard
for an upheaval to completely wipe out every last vestige of life on the
planet. And a single small colony,
buried deep in the mud at the bottom of a deep ocean, would be enough to
re-colonise the planet as soon as conditions eased.
This picture of an Achaean landscape is by Peter
Sawyer of the Smithsonian Institute.
The Isua rocks that we mentioned above are something
under 4 thousand million years old (3.8 kMy I think is
the official figure). And they contain
tantalising deposits of carbon. Now
carbon deposits are normally the remains of long-dead life (think coal). But ‘extraordinary claims require
extraordinary evidence’, and many scientists regard the idea that life could
have got going so early as truly extraordinary.
I can’t see what’s extraordinary about it myself, but there you go. Huge amounts of research have gone into
trying to prove beyond any shadow of doubt that the Isua carbon is indeed the
dead remains of early life. And the
story has fluctuated wildly down the years.
I think we can safely say that life ‘may have’ been
around as early as this. Or even that it
‘probably was’. But we may never be able
to say that it ‘certainly was’. My guess
is that, if there was life around at Isua time, then it was probably a
primitive form of something not unlike today’s bacteria.
The first solid evidence
of Life is ‘only’ 3½ thousand million years ago. And I’ve read that some scientists dispute
even that.
The evidence takes the form of ‘micro’ fossils. As the link shows, they look remarkably like
modern bacteria – so like in fact that the scientists that discovered them even
gave them pseudo modern names. Bacteria
may be one of the 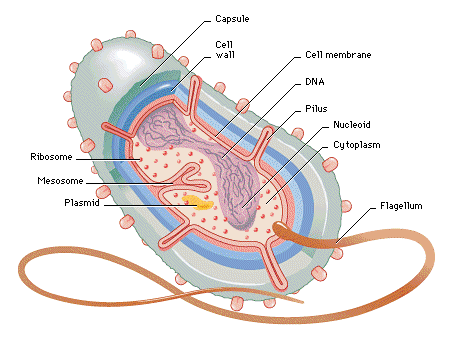 simplest
life-forms on the planet. But they are
very sophisticated little beasties just the same. I’ve put this diagram in just to show
this. You will have to look on the
Internet if you want to know what the various components are.
simplest
life-forms on the planet. But they are
very sophisticated little beasties just the same. I’ve put this diagram in just to show
this. You will have to look on the
Internet if you want to know what the various components are.
Some bacteria can move. In the jargon, they are ‘motile’ (what’s
wrong with ‘mobile’ you may ask, and I have no answer). This is a motile one. The small lump on the end of the tail is
called a proton motor, and it spins. The
spinning tail propels the bacterium in a haphazard way, hopefully away from
somewhere it doesn’t want to be to somewhere more desirable. This diagram only depicts one
‘flagellum’. But many pictures of
bacteria, including the one in bacteria, show them
sporting a forest of them.
By the time anything like this appears in the fossil
record, life was already highly developed, and must have been going for a long
time. More
How did Life emerge?
We’ll probably never know. It all happened a very long time ago, on an
Earth substantially different from our modern one. Scientists are struggling to understand how
emerging life could have made the jump from simple chemicals to even the
simplest modern life-form. As the
picture shows, even the humble bacterium is actually a very sophisticated
organism. Viruses don’t count. They are pure parasites, and can’t exist
without a more advanced host to prey on.
But there are some things that we can say to make it
all seem a little less miraculous.
 First
the process will not have been simple (is it ever in this game?). There will have been many false starts and
many side alleys travelled down.
First
the process will not have been simple (is it ever in this game?). There will have been many false starts and
many side alleys travelled down.
An increasingly popular theory was originated by one
Gunther Wächtershäuser. Wächtershäuser
believes that the key ingredient that enabled life to get started was pyrite
(iron pyrites, fool’s gold). You can see
from the picture why it’s called fool’s gold.
The yellow specks are very realistic.
Pyrite is a mixture of crystals of hydrogen sulphide (H2S,
bad-eggs smell) and iron sulphide (FeS).
And it grows spectacularly around the hydrothermal vents in the deep
ocean.
 We
won’t go into it. It’s on the Internet
(don’t forget the first ‘s’ if you look Wächtershäuser up). But it seems that various organic compounds
are generated naturally, by processes that are entirely ‘inorganic’.
We
won’t go into it. It’s on the Internet
(don’t forget the first ‘s’ if you look Wächtershäuser up). But it seems that various organic compounds
are generated naturally, by processes that are entirely ‘inorganic’.
To get far, any emerging life-form would have to have
some kind of enclosure, so that it doesn’t lose any building blocks. I’ve seen a number of ideas about what the
very first enclosure might have been.
But it seems that the ‘prebiotic soup’ will have contained a plentiful
supply of detergent molecules. These
detergent molecules form naturally into underwater bubbles, as this diagram
from Scientific American (Oct. ’85)
shows.
The blue-blob end loves water, but the the tail is
waxy and hates it. If you pour some
detergent into water then it will automatically form itself into bubbles, with
the water-loving end facing out, and the other end protected.
But that’s not all.
The bubble wall is automatically ‘semi-permeable’. It will allow the small molecules of simple
chemicals through. But if these simple
chemicals should combine together to form larger molecules – either by chance
or because there’s some deliberate chemistry going on – then they cannot get
out again. They are trapped. If the bubble should get too large and burst,
the edges immediately close up again, to form two new offspring.
Emerging life took advantage of this natural feature
of 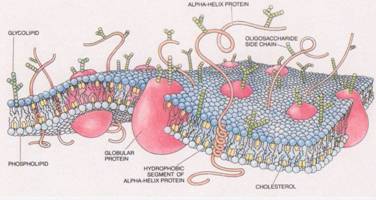 detergent
bubbles. So the cells walls of bacteria
are basically made of soap – as are the cells that you are made of. Our cells are much more sophisticated, as the
second picture shows. But you can still
see the soap molecules holding it all together.
detergent
bubbles. So the cells walls of bacteria
are basically made of soap – as are the cells that you are made of. Our cells are much more sophisticated, as the
second picture shows. But you can still
see the soap molecules holding it all together.
The next thing that emerging life needs is some kind
of ‘blueprint’ from which to make its chemical building blocks. Modern life uses DNA. DNA is ideal for the job, but it’s a
sophisticated molecule and difficult to make.
The RNA, that waits upon it like a servant and does its bidding, is less
good as a permanent record. But it’s
simpler, and there are hints that it may have done the job on its own at one
time. Before that, who knows?
The third thing that life needs is energy. The ideal energy source for life, even
today, is glucose (C6H12O6). Amazingly, it seems that there was glucose
around in the ‘pre-biotic soup’.
Experiments to simulate the early-earth environment produce it, together
with a wide range of other sugars. I got
this fact from William Schopf’s book
‘Cradle of Life’, so how it fits in with Wächtershäuser’s work I can’t tell
you.
And finally, life needs raw materials. The raw materials of life boil down to just
four elements, what Schopf calls CHON – Carbon, Hydrogen, Oxygen and
Nitrogen. The first three were already
available in the sugar. Emerging life
will have had plenty of ammonia available (NH3), which which will
have given it the nitrogen.
On this page we have been into speculation, and trying
to establish what might have been possible.
The next chapter in the tale involves organisms very
much like modern bacteria.
© C B Pease, March 08